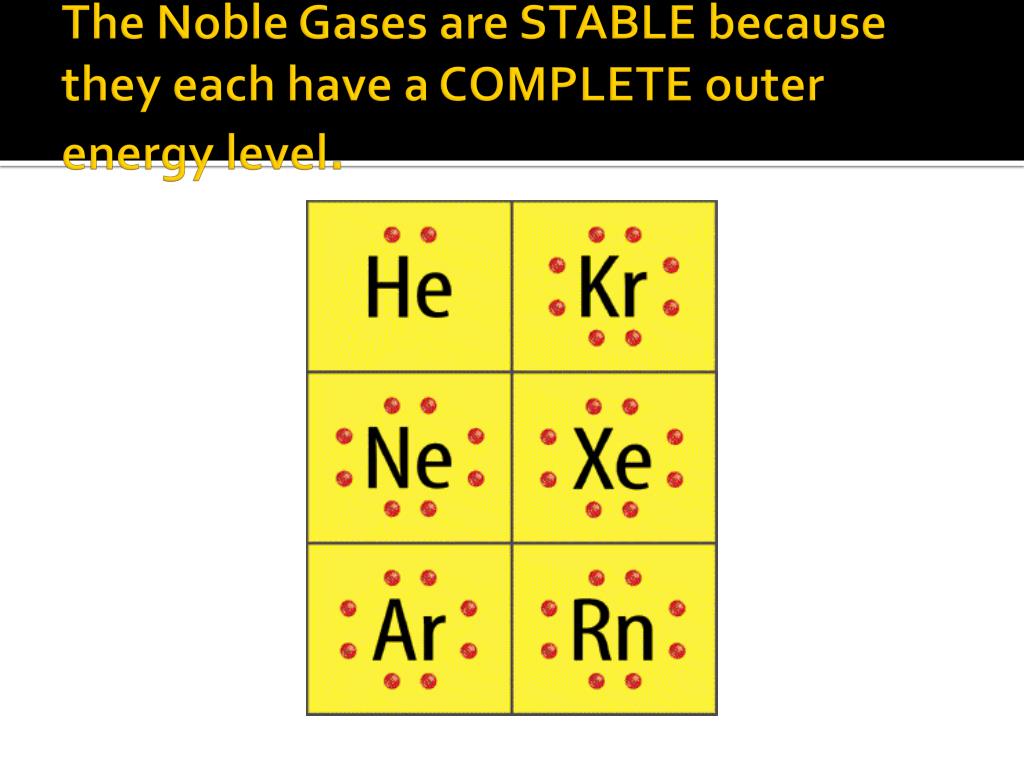
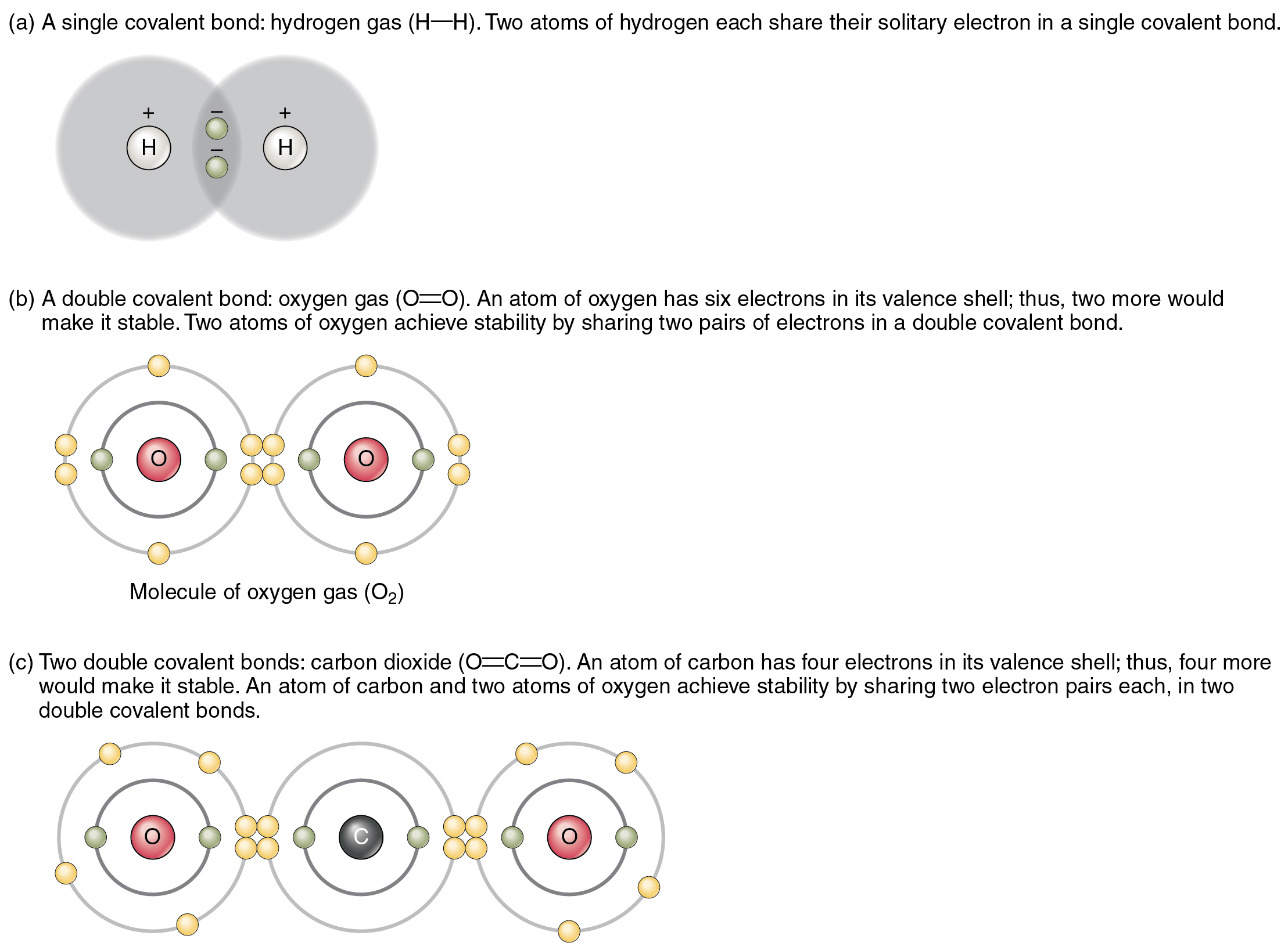
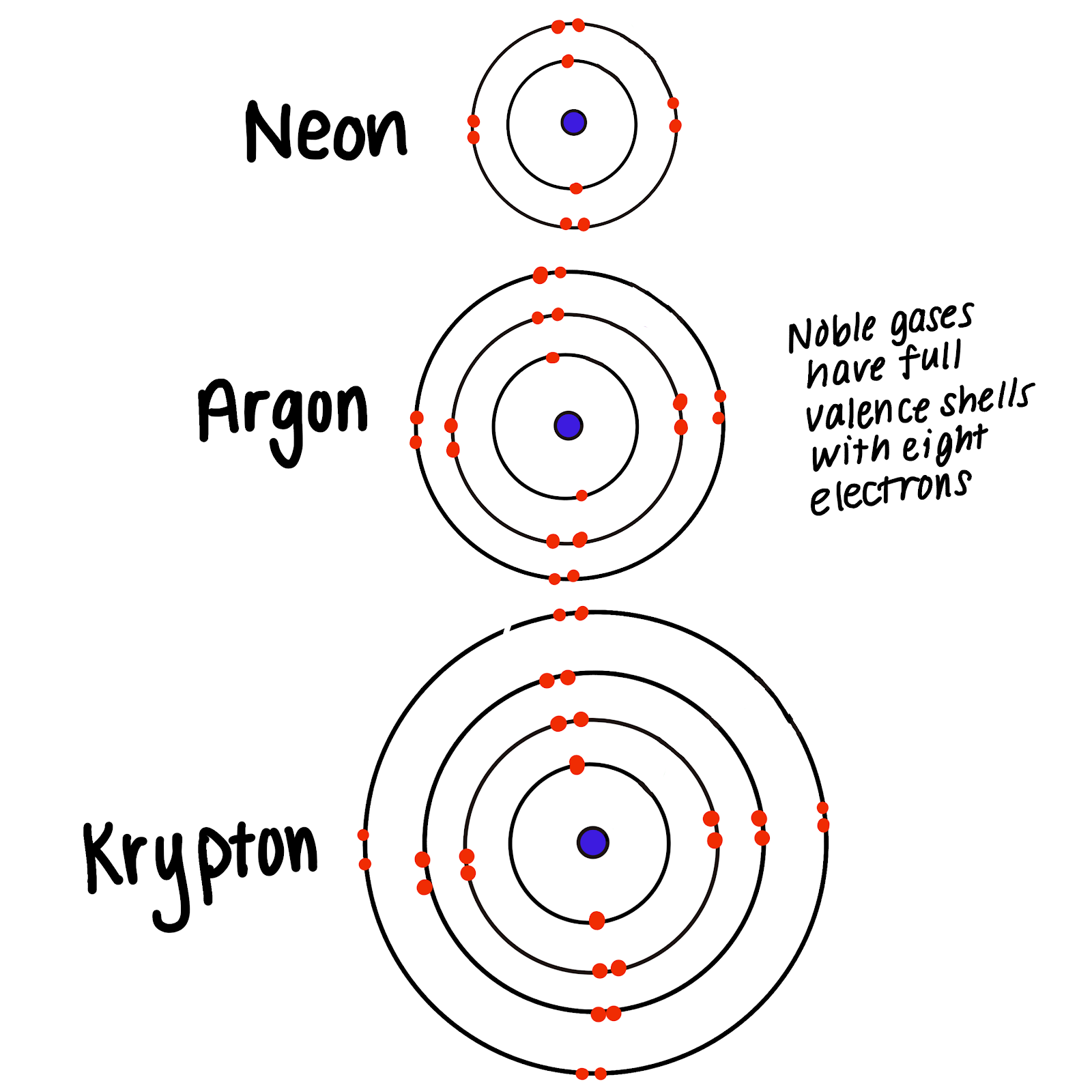
Why Are Noble Gases Not Likely To Form Chemical Bonds
Why Are Noble Gases Not Likely To Form Chemical Bonds - In fact, they would lose something and likely have to go to a higher energy state in order to form a bond. Another popular term is “noble gases,” suggesting that. Web therefore they're not going to gain anything by forming bonds. Ad over 27,000 video lessons and other resources, you're guaranteed to find what you need. Web explain why noble gases usually do not form bonds with other atoms? Atoms that will form chemical bonds are referred to as ____ , whereas. Web they traditionally have been labeled group 0 in the periodic table because for decades after their discovery it was believed that they could not bond to other. The full valence electron shells of these atoms make. Web noble gases are odorless, colorless, nonflammable, and monotonic gases that have low chemical reactivity. Web noble gases elements are located in group 18 and known for their general electron configuration of n s 2 n p 6 ns^2 np^6 n s 2 n p 6 (with the exception of helium) which. Because they already have an electron configuration with a full, stable outer energy level. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Noble gases (ngs) are the least reactive elements in the periodic table towards chemical bond formation when compared with. Web because of the stability in their electronic configuration, nobel gases do not share its outermost electron with any other atom. Web because noble gases’ outer shells are full, they are extremely stable, tending not to form chemical bonds and having a small tendency to gain or lose. Web this group has been referred to as the “inert” gases, indicating. The full valence electron shells of these atoms make. In fact, they would lose something and likely have to go to a higher energy state in order to form a bond. Noble gases (ngs) are the least reactive elements in the periodic table towards chemical bond formation when compared with other elements because of. Web explain why noble gases usually. And in this form we can say that they do not. Web because of the stability in their electronic configuration, nobel gases do not share its outermost electron with any other atom. Web noble gases elements are located in group 18 and known for their general electron configuration of n s 2 n p 6 ns^2 np^6 n s 2. Atoms that will form chemical bonds are referred to as ____ , whereas. Web therefore they're not going to gain anything by forming bonds. The full valence electron shells of these atoms make. Web explain why noble gases usually do not form bonds with other atoms? Web noble gases are located next to the halogen group in the periodic table. Web explain why noble gases usually do not form bonds with other atoms? Because they’re reluctant to share electrons from their filled outer electron shells, noble gases are. Web noble gases are odorless, colorless, nonflammable, and monotonic gases that have low chemical reactivity. Web they traditionally have been labeled group 0 in the periodic table because for decades after their. Because they’re reluctant to share electrons from their filled outer electron shells, noble gases are. Web explain why noble gases usually do not form bonds with other atoms? The full valence electron shells of these atoms make. Web noble gases are odorless, colorless, nonflammable, and monotonic gases that have low chemical reactivity. Web noble gases elements are located in group. Web explain why noble gases usually do not form bonds with other atoms? Web explain why noble gases are not likely to form chemical bonds. Web they traditionally have been labeled group 0 in the periodic table because for decades after their discovery it was believed that they could not bond to other. Web instead, one might describe the noble. Web instead, one might describe the noble gases as aloof. Mhm bonds form in order for atoms to achieve a lower energy state, they achieve a lower energy state by simultaneously achieving and a noble gas electron. Web therefore they're not going to gain anything by forming bonds. Atoms that will form chemical bonds are referred to as ____ ,. Web because of the stability in their electronic configuration, nobel gases do not share its outermost electron with any other atom. Web because noble gases’ outer shells are full, they are extremely stable, tending not to form chemical bonds and having a small tendency to gain or lose. Web therefore they're not going to gain anything by forming bonds. In. Because they already have an electron configuration with a full, stable outer energy level. Web this group has been referred to as the “inert” gases, indicating that they are chemically inert, or unreactive. Web noble gases are odorless, colorless, nonflammable, and monotonic gases that have low chemical reactivity. Web therefore they're not going to gain anything by forming bonds. The full valence electron shells of these atoms make. Web instead, one might describe the noble gases as aloof. Web explain why noble gases usually do not form bonds with other atoms? Web because noble gases’ outer shells are full, they are extremely stable, tending not to form chemical bonds and having a small tendency to gain or lose. Web noble gases elements are located in group 18 and known for their general electron configuration of n s 2 n p 6 ns^2 np^6 n s 2 n p 6 (with the exception of helium) which. Another popular term is “noble gases,” suggesting that. Web all noble gases have full s and p outer electron shells (except helium, which has no p sublevel), and so do not form chemical compounds easily. Mhm bonds form in order for atoms to achieve a lower energy state, they achieve a lower energy state by simultaneously achieving and a noble gas electron. Ad over 27,000 video lessons and other resources, you're guaranteed to find what you need. Web because of the stability in their electronic configuration, nobel gases do not share its outermost electron with any other atom. Atoms that will form chemical bonds are referred to as ____ , whereas. And in this form we can say that they do not. In fact, they would lose something and likely have to go to a higher energy state in order to form a bond. Noble gases (ngs) are the least reactive elements in the periodic table towards chemical bond formation when compared with other elements because of. Web explain why noble gases are not likely to form chemical bonds. Web they traditionally have been labeled group 0 in the periodic table because for decades after their discovery it was believed that they could not bond to other.What Are Noble Gases? Definition and Properties
PPT Recap Atomic Structure PowerPoint Presentation, free download
Why Do Noble Gases Not React WHYPLJ
PPT Chapter 19— CHEMICAL BONDS PowerPoint Presentation, free download
How to Write a Noble Gas Configuration for Atoms of an Element
Why Atoms Make Bonds Why Noble Gases are Stable Chemical Bonding
Chemical Bonds · Anatomy and Physiology
Noble Gas Configuration For Silicon Schott Gromentiout
Describe the Properties of Noble Gases
PPT Introduction to the Periodic Table PowerPoint Presentation, free
Related Post:

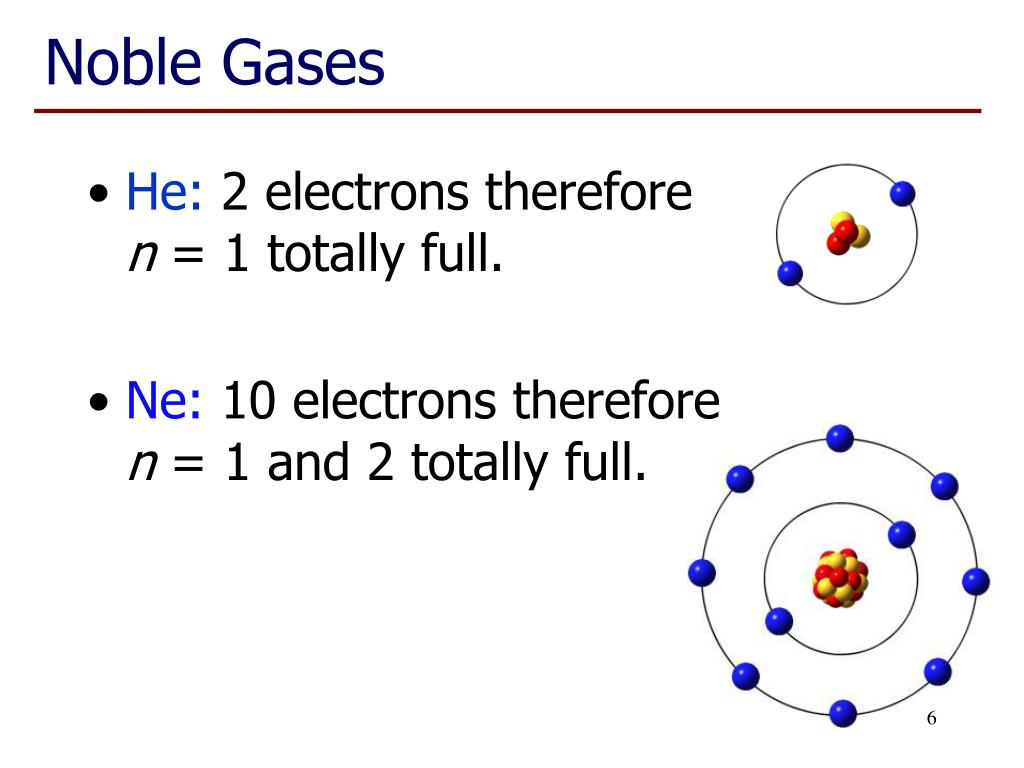
.PNG)