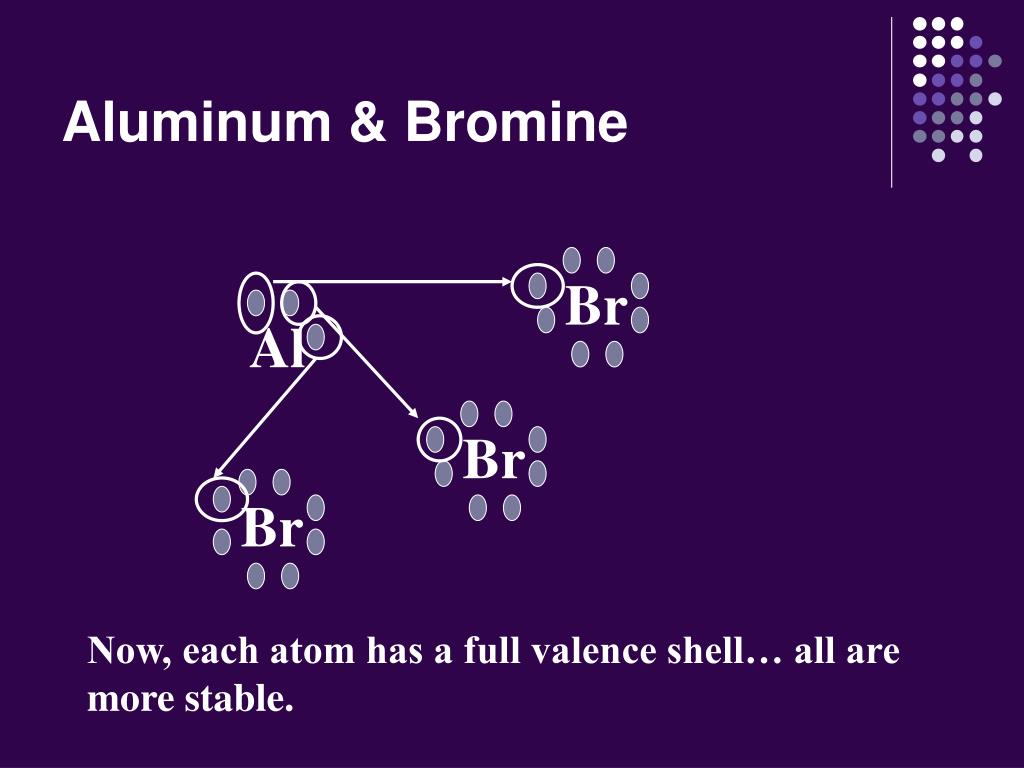
How Many Bonds Can Bromine Form
How Many Bonds Can Bromine Form - Web there is a single bond with bromine atoms and three lone pairs on each bromine atoms. Group 5a form 3 bonds; A double bond is formed when two atoms use two electron pairs to form two covalent. Web typically, the atoms of group 4a form 4 covalent bonds; And group 7a form one bond. Web in bromine's case, it only lacks 1 electron to have a complete octet, so it will ty vigurously to get that one electron. Ad enjoy great deals and discounts on an array of products from various brands. The maximum number of bonds. Web typically, the atoms of group 4a form 4 covalent bonds; Web let's illustrate how a covalent bond forms between iodine and bromine, with the understanding that each atom only needs one more electron to complete an octet Group 5a form 3 bonds; A double bond is formed when two atoms use two electron pairs to form two covalent. This can be seen from the standard electrode potentials of the x2/x couples (f, +2.866 v; Bromine, which belongs to group 17 and period four of the periodic table, has seven outer shell or valence. Web science chemistry how. Web typically, the atoms of group 4a form 4 covalent bonds; This can be seen from the standard electrode potentials of the x2/x couples (f, +2.866 v; View the full answer transcribed image text: Bromine, which belongs to group 17 and period four of the periodic table, has seven outer shell or valence Web if either iodine or bromine were. So, this lewis structure is a very simple. Group 5a form 3 bonds; Web bromine will normally form one covalent bond. Bromine, which belongs to group 17 and period four of the periodic table, has seven outer shell or valence. Web under certain bonding conditions, adjacent atoms will form multiple bonds with each other. View the full answer transcribed image text: To do that, a bromine atom forms a covalent bond. Web bromine will normally form one covalent bond. Web in bromine's case, it only lacks 1 electron to have a complete octet, so it will ty vigurously to get that one electron. Web expert answer 100% (6 ratings) carbon atom has four valence. Web typically, the atoms of group 4a form 4 covalent bonds; * a) 1 b) 2 c) 3 d) 4 e) 7 how many bonds does bromine atoms form in. Group 6a form 2 bonds; Web in bromine's case, it only lacks 1 electron to have a complete octet, so it will ty vigurously to get that one electron. Bromine,. Get deals and low prices on bromine mineral at amazon View the full answer transcribed image text: Modeling lonic and covalent bonds part 1: Group 5a form 3 bonds; Group 6a form 2 bonds; It’s called the honc rule, or sometimes known as honc 1234 rule. A double bond is formed when two atoms use two electron pairs to form two covalent. Hydrogen makes 1 bond, oxygen makes 2 bonds, nitrogen makes. Web typically, the atoms of group 4a form 4 covalent bonds; Web if either iodine or bromine were to given up valence. Group 6a form 2 bonds; Group 6a form 2 bonds; Web the atomic number of al is 13, and its electronic configuration is 1s22p22p63s23p1. Group 5a form 3 bonds; The maximum number of bonds. View the full answer transcribed image text: Hydrogen makes 1 bond, oxygen makes 2 bonds, nitrogen makes. Web science chemistry how many bonds does bromine atoms form in neutral organic compounds? 1 bond how many bonds can sulfur form with neighboring atoms in a compound? Web how many bonds can bromine form with neighboring atoms in a compound? Group 5a form 3 bonds; Web expert answer 100% (6 ratings) carbon atom has four valence electr. The number refers to the number of bonds each of the element makes: Bromine is intermediate in reactivity between chlorine and iodine, and is one of the most reactive elements. Group 5a form 3 bonds; Bromine, which belongs to group 17 and period four of the periodic table, has seven outer shell or valence. Web under certain bonding conditions, adjacent atoms will form multiple bonds with each other. Group 5a form 3 bonds; So, this lewis structure is a very simple. It’s called the honc rule, or sometimes known as honc 1234 rule. Web expert answer 100% (6 ratings) carbon atom has four valence electr. Hydrogen makes 1 bond, oxygen makes 2 bonds, nitrogen makes. This can be seen from the standard electrode potentials of the x2/x couples (f, +2.866 v; When bromine atoms form covalent bonds with other atoms,. Web if either iodine or bromine were to given up valence electrons to form a cation, they would have to give up all seven valence electrons to reveal the next Web let's illustrate how a covalent bond forms between iodine and bromine, with the understanding that each atom only needs one more electron to complete an octet Ad enjoy great deals and discounts on an array of products from various brands. Group 5a form 3 bonds; Modeling lonic and covalent bonds part 1: The number refers to the number of bonds each of the element makes: Web this comes in handy especially when drawing lewis structures. Web bromine will normally form one covalent bond. * a) 1 b) 2 c) 3 d) 4 e) 7 how many bonds does bromine atoms form in. Group 6a form 2 bonds; Web bromine will normally form one covalent bond.Allylic Bromination by NBS with Practice Problems Chemistry Steps
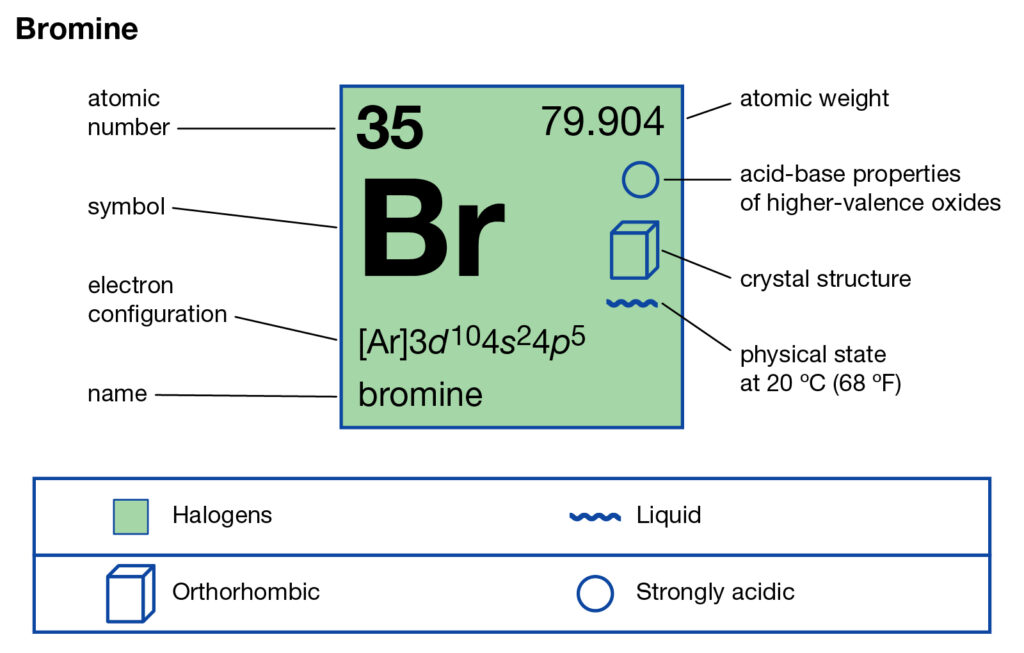
Bromine Periodic Table and Atomic Properties
Bromine Formula Symbol of Bromine, Density, Charge Embibe
How to predict number of bonds each element makes Predictions
How Can We Find A Electron Configuration For Bromine (Br)
PPT Chemical Bonding Bonding Theory and Lewis Formulas PowerPoint
How Many Valence Electrons Does Bromine (Br) Have? [Valency of Bromine]
PPT Chemistry Unit 2 PowerPoint Presentation, free download ID3668529
Bromine Valence Electrons Bromine Valency (Br) Dot Diagram
How Can We Find A Electron Configuration For Bromine (Br)
Related Post:






![How Many Valence Electrons Does Bromine (Br) Have? [Valency of Bromine]](https://1.bp.blogspot.com/-ynLTJPSFkNY/YBXNXDx8ACI/AAAAAAAADZU/h1jbYW5duhMr7iiZUWhxGFBBUoDEUho1wCLcBGAsYHQ/w1600/Screenshot%2B%252878%2529-min.png)